Oral methylene blue has not been approved for the treatment of any condition
and where used, represents off-label use of an FDA-grandfathered pharmaceutical.
The following have been selectively curated and highlighted; reader discretion advised. Links provided to source material for further reading where identified.
While these abstracts suggest a basis for use of methylene blue in some conditions, their relevance to any specific individual can only be determined in the context of a physician patient relationship.
A controlled trial of methylene blue in severe depressive illness
GJ Naylor, A H Smith, P Connelly
PMID: 3555627 DOI: 10.1016/0006-3223(87)90194-6
Abstract
Methylene blue, 15 mg/day, was compared with placebo in treatment of severe depressive illness. The 3-week trial was designed to avoid bias by placebo response and also to avoid observer bias. Improvement in patients receiving methylene blue was significantly greater than in those receiving placebo. Methylene blue at a dose of 15 mg/day appears to be a potent antidepressant, and further clinical evaluation is essential.
Effects of post-session administration of methylene blue on fear extinction and contextual memory in adults with claustrophobia
Michael J Telch, Aleksandra K Bruchey, David Rosenfield, Adam R Cobb, Jasper Smits, Sandra Pahl, F Gonzalez-Lima
PMID: 25018057 PMCID: PMC4467026 DOI: 10.1176/appi.ajp.2014.13101407
Free PMC article
Abstract
Objective: Preclinical studies have shown that low-dose methylene blue increases mitochondrial cytochrome oxidase activity in the brain and improves memory retention after learning tasks, including fear extinction. The authors report on the first controlled experiment to examine the memory-enhancing effects of post training methylene blue administration on retention of fear extinction and contextual memory following fear extinction training.
Method: Adult participants displaying marked claustrophobic fear were randomly assigned to double-blind administration of 260 mg of methylene blue (N=23) or administration of placebo (N=19) immediately following six 5-minute extinction trials in an enclosed chamber. Retesting occurred 1 month later to assess fear renewal as indexed by peak fear during exposure to a nontraining chamber, with the prediction that the effects of methylene blue would vary as a function of fear reduction achieved during extinction training. Incidental contextual memory was assessed 1 and 30 days after training to assess the cognitive-enhancing effects of methylene blue independent of its effects on fear attenuation.
Results: Consistent with predictions, participants displaying low end fear post training showed significantly less fear at the 1-month follow-up if they received methylene blue post training compared with placebo. In contrast, participants displaying moderate to high levels of post training fear tended to fare worse at the follow-up if they received methylene blue post training. Methylene blue's enhancement of contextual memory was unrelated to initial or post training claustrophobic fear.
Conclusions: Methylene blue enhances memory and the retention of fear extinction when administered after a successful exposure session but may have a deleterious effect on extinction when administered after an unsuccessful exposure session.
Enhancing Extinction Learning in Posttraumatic Stress Disorder With Brief Daily Imaginal Exposure and Methylene Blue: A Randomized Controlled Trial
Lori A Zoellner 12, Michael Telch 3, Edna B Foa*, Frank J Farach 2, Carmen P McLean, Robert Gallop 3, Ellen J Bluett, Adam Cobb 3, F Gonzalez-Lima 3
Affiliations + expand
PMID: 28686823 DOE 10.4088/JCP.16m10936
Abstract
Objective: The memory-enhancing drug methylene blue (MB) administered after extinction training improves fear extinction retention in rats and humans with claustrophobia. Robust findings from animal research, in combination with established safety and data showing MB-enhanced extinction in humans, provide a foundation to extend this work to extinction-based therapies for posttraumatic stress disorder (PTSD) such as prolonged exposure (PE).
Methods: Patients with chronic PTSD (DSM-IV-TR; N = 42) were randomly assigned to imaginal exposure plus MB (IE + MB), imaginal exposure plus placebo (IE + PBO), or waitlist (WL/standard PE) from September 2011 to April 2013. Following 5 daily, 50-minute imaginal exposure sessions, 260 mg of MB or PBO was administered. Waitlist controls received PE following 1-month follow-up. Patients were assessed using the independent evaluator-rated PTSD Symptom Scale-Interview version (primary outcome), patient-rated PTSD, trauma-related psychopathology, and functioning through 3-month follow-up.
Results: Both IE + MB and IE + PBO showed strong clinical gains that did not differ from standard PE at 3-month follow-up. MB-augmented exposure specifically enhanced independent evaluator-rated treatment response (number needed to treat = 7.5) and quality of life compared to placebo (effect size d=0.58). Rate of change for IE + MB showed a delayed initial response followed by accelerated recovery, which differed from the linear pattern seen in IE + PBO, MB effects were facilitated by better working memory but not by changes in beliefs.
Conclusions: The findings provide preliminary efficacy for a brief IE treatment for PTSD and point to the potential utility of MB for enhancing outcome. Brief interventions and better tailoring of MB augmentation strategies, adjusting for observed patterns, may have the potential to reduce dropout, accelerate change, and improve outcomes.
A two-year double-blind crossover trial of the prophylactic effect of methylene blue in manic- depressive psychosis
GJ Naylor, B Martin, SE Hopwood, Y Watson
PMID: 3091097 DOE 10.1016/0006-3223(86)90265-9
Abstract
A 2-year prophylactic trial was carried out in 31 bipolar manic-depressive subjects, comparing 300 mg/day methylene blue on a double-blind crossover basis with 15 mg/day. All patients were also maintained on lithium. Seventeen patients completed the 2-year trial. During the year the patients were treated with methylene blue at 300 mg/day, they were significantly less depressed than during the year on 15 mg/day. No significant difference in the severity of manic symptoms was shown. The trial had obvious limitations, e.g., a small number of subjects, a relatively large number of dropouts, relatively simple rating scales, doubts about blindness, and uncertainty as to whether or not 15 mg methylene blue per day could be considered a placebo. However, the results suggest that methylene blue may be a useful addition to lithium in the long-term treatment of manic-depressive psychosis and warrants further investigation.
Methylene blue adjuvant therapy of schizophrenia
SI Deutsch, RB Rosse, B L. Schwartz, M Fay-McCarthy, P B Rosenberg, K Fearing
Affiliations + expand
PMID: 9260734 DOI: 10.1097/00002826-199708000-00008
Abstract
There is growing interest in the role of the nitric oxide (NO) pathway in idiopathic psychotic disorders such as schizophrenia. In this preliminary study, we examined the therapeutic efficacy of methylene blue (MB), a "downstream" inhibitor of one of NO's actions, administered orally as an adjuvant to conventional neuroleptic medications. Specifically, MB blocks NO's activation of soluble guanylyl cyclase. MB has previously been reported to have therapeutic effects in the treatment of psychosis and mania. Preclinical data also suggest that MB might possess antipsychotic potential. Participants in the current study were eight patients with schizophrenia who had incomplete responses to conventional antipsychotics (as evidenced by a Brief Psychiatric Rating Scale [BPRS] total score of 35 or more). These patients completed a 4-week open-label study with a 1 week "off", 2 week "on", and one final week "off" design. Measures of treatment efficacy were the BPRS, Schedule for the Assessment of Negative Symptoms, and Clinical Global Improvement Scale administered weekly. Final scores for each outcome measure item were based on the consensus of at least two trained raters present during each rating interview. A statistically significant, albeit modest, decrease in the severity of psychopathology was observed while the subjects were taking MB, and psychopathology significantly worsened when MB was discontinued. The results suggest a need for further study with MB or perhaps other NO-dependent guanylyl cyclase-inhibiting medications.
Methylene blue modulates functional connectivity in the human brain
Pavel Rodriguez 12, Amar P Singh 3, Kristen E Malloy, Wei Zhou, Douglas W Barrett 5, Crystal G Franklin 4, Wilson B Altmeyer 3, Juan E Gutierrez 3, Jinqi Li 4, Betty L Heyl * Jack L Lancaster 4, F Gonzalez-Lima 5, Timothy Q Duong
Affiliations + expand
PMID: 26961091 PMCID: PMC5018244 DOI: 10.1007/s11682-016-9541-6
Free PMC article
Abstract
Methylene blue USP (MB) is a FDA-grandfathered drug used in clinics to treat methemoglobinemia, carbon monoxide poisoning and cyanide poisoning that has been shown to increase fMRI evoked blood oxygenation level dependent (BOLD) response in rodents. Low dose MB also has memory enhancing effect in rodents and humans. However, the neural correlates of the effects of MB in the human brain are unknown. We tested the hypothesis that a single low oral dose of MB modulates the functional connectivity of neural networks in healthy adults. Task-based and task-free fMRI were performed before and one hour after MB or placebo administration utilizing a randomized, double-blinded, placebo-controlled design. MB administration was associated with a reduction in cerebral blood flow in a task-related network during a visuomotor task, and with stronger resting-state functional connectivity in multiple regions linking perception and memory functions. These findings demonstrate for the first time that low-dose MB can modulate task-related and resting-state neural networks in the human brain. These neuroimaging findings support further investigations in healthy and disease populations.
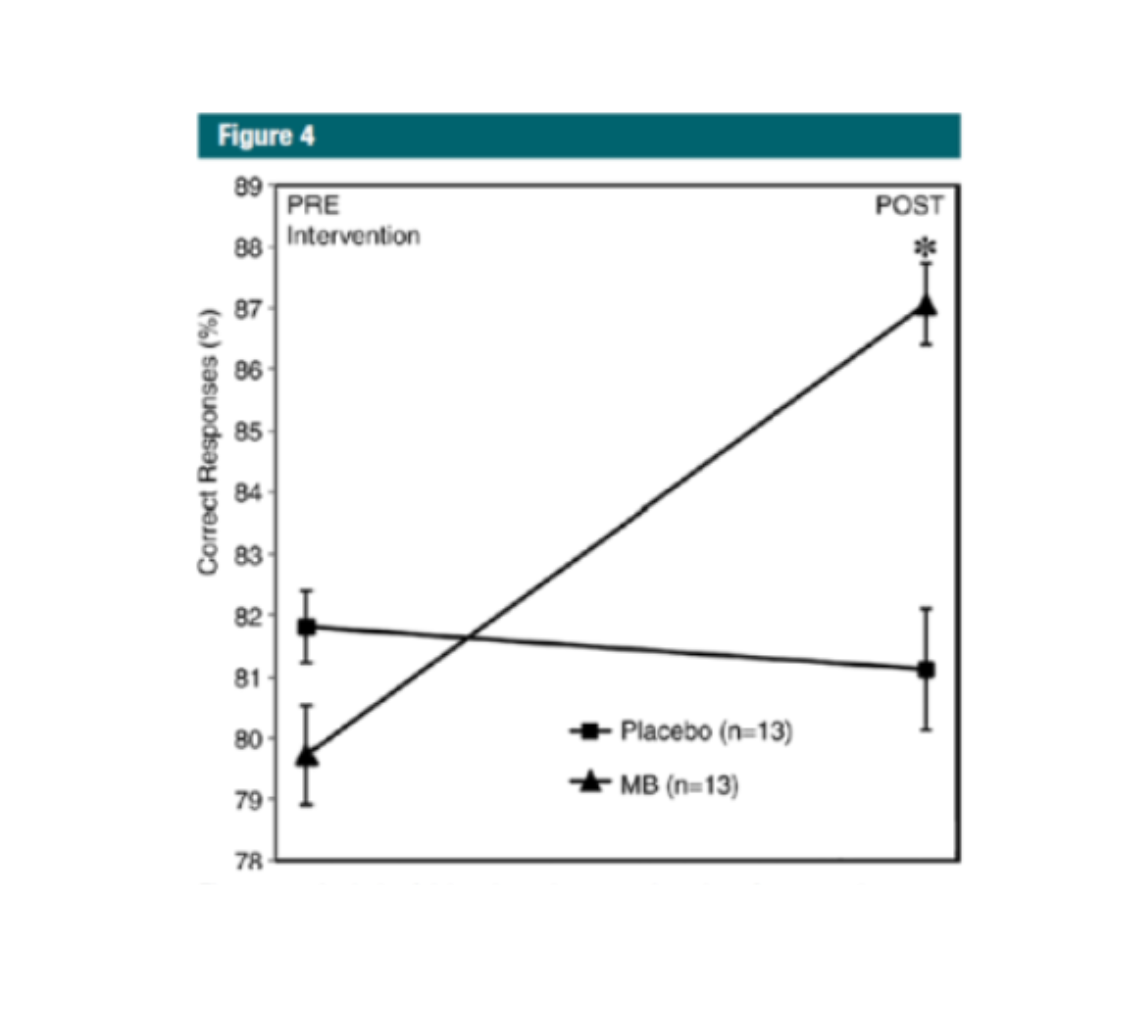
Figure 4: Analysis of delayed match-to-sample task performance change (after intervention to before intervention) during the retrieval phase shows a significant increase in correct responses only in the methylene blue (MB) group (P=.01). Error bars = standard error of mean.
as a surgical stain (2). The pharma- cokinetic and side effects of low-dose (0.5-4.0 mg/kg) methylene blue are well known and minimal in humans
(3,4). When the lipophilic methylene blue enters the mitochondria of the brain at low concentrations, it forms a renewable redox complex that can do- nate electrons to the electron transport chain, thereby promoting cytochrome c oxidase activity, oxygen consumption, and energy production (5-7). While
Purpose:
Materials and Methods:
Results:
Conclusion:
To investigate the sustained-attention and memory-en- hancing neural correlates of the oral administration of methylene blue in the healthy human brain.
The institutional review board approved this prospective, HIPAA-compliant, randomized, double-blinded, placebo- controlled clinical trial, and all patients provided informed consent. Twenty-six subjects (age range, 22-62 years) were enrolled. Functional magnetic resonance (MR) im- aging was performed with a psychomotor vigilance task (sustained attention) and delayed match-to-sample tasks (short-term memory) before and 1 hour after adminis- tration of low-dose methylene blue or a placebo. Cere- brovascular reactivity effects were also measured with the carbon dioxide challenge, in which a 2 × 2 repeated- measures analysis of variance was performed with a drug (methylene blue vs placebo) and time (before vs after ad- ministration of the drug) as factors to assess drug X time between group interactions. Multiple comparison correc- tion was applied, with cluster-corrected P<.05 indicating a significant difference.
Administration of methylene blue increased response in the bilateral insular cortex during a psychomotor vigilance task (Z=2.9-3.4, P = .01-.008) and functional MR imag- ing response during a short-term memory task involving the prefrontal, parietal, and occipital cortex (Z = 2.9-4.2, P=.03-0003). Methylene blue was also associated with a 7% increase in correct responses during memory retrieval (P = .01).
Low-dose methylene blue can increase functional MR im- aging activity during sustained attention and short-term memory tasks and enhance memory retrieval.
*RSNA, 2016
Online supplemental material is available for this article.






